Elektronische Signaturen gemäß 21 CFR Part 11






Verständnis von 21 CFR Teil 11 und seiner Rolle bei elektronischen Signaturen
In regulierten Branchen wie Pharma, Biotechnologie und Medizintechnik ist die Sicherstellung der Einhaltung von Standards für elektronische Signaturen entscheidend für die Aufrechterhaltung der Datenintegrität und die Vorbereitung auf Audits. 21 CFR Teil 11 ist eine von der US-amerikanischen Food and Drug Administration (FDA) erlassene Verordnung, die die Kriterien festlegt, nach denen elektronische Aufzeichnungen und elektronische Signaturen als vertrauenswürdig, zuverlässig und gleichwertig mit Papieraufzeichnungen und handschriftlichen Unterschriften gelten. Dieser Rahmen entstand in den späten 1990er Jahren als Reaktion auf die zunehmende Verbreitung digitaler Technologien im FDA-regulierten Umfeld, um Probleme wie Datenmanipulation oder unbefugten Zugriff zu verhindern.
Im Kern erfordert die Einhaltung von 21 CFR Teil 11 die Implementierung von Kontrollmaßnahmen durch Systeme, um die Authentizität elektronischer Signaturen zu überprüfen. Dazu gehören eindeutige Benutzeridentifikation, sichere Zugriffskontrollen, Audit Trails, die alle Aktionen aufzeichnen, und Mechanismen, die sicherstellen, dass Signaturen nicht gefälscht oder geleugnet werden können. Für Unternehmen, die in den USA tätig sind, insbesondere solche, die Daten an die FDA übermitteln, kann die Nichteinhaltung schwerwiegende Folgen haben, wie z. B. Verzögerungen bei der Produktzulassung, Geldstrafen oder rechtliche Haftung. Aus geschäftlicher Sicht mindert die Einführung einer 21 CFR Teil 11-konformen Lösung für elektronische Signaturen nicht nur Risiken, sondern rationalisiert auch Arbeitsabläufe und reduziert die Abhängigkeit von fehleranfälligen und zeitaufwändigen manuellen Prozessen.
Die Verordnung gilt umfassend für elektronische Aufzeichnungen in Bereichen wie klinische Studien, Fertigungsqualitätskontrolle und regulatorische Einreichungen. Unternehmen müssen Anbieter von elektronischen Signaturen anhand dieser Standards bewerten, einschließlich Funktionen wie Zeitstempel, Verschlüsselung und Authentifizierung der Unterzeichneridentität. Mit der Beschleunigung der digitalen Transformation ist die Nachfrage nach solchen Compliance-Tools sprunghaft angestiegen, und Marktanalysten prognostizieren ein stetiges Wachstum des Marktes für elektronische Signaturen aufgrund des regulatorischen Drucks.

Hauptanforderungen für 21 CFR Teil 11-konforme elektronische Signaturen
Um die 21 CFR Teil 11-Konformität zu erreichen, müssen elektronische Signaturplattformen mehrere technische und verfahrenstechnische Schutzmaßnahmen integrieren. Erstens müssen elektronische Signaturen so mit der entsprechenden Aufzeichnung verknüpft sein, dass Änderungen nach der Unterzeichnung verhindert werden. Dies beinhaltet typischerweise digitale Zertifikate und eine Public-Key-Infrastruktur (PKI), um die Identität des Unterzeichners zu überprüfen. Zweitens müssen Systeme detaillierte Audit Trails erstellen, die erfassen, wer wann was unterzeichnet hat, und die Unveränderlichkeit durch Blockchain-ähnliche Protokolle oder sichere Datenbanken gewährleisten.
Validierung ist eine weitere Säule: Plattformen müssen strengen Tests unterzogen werden, um ihren konsistenten Betrieb unter verschiedenen Bedingungen zu bestätigen, einschließlich Systemausfällen oder Cyber-Bedrohungen. Die FDA betont die Unterscheidung zwischen "offenen Systemen" und "geschlossenen Systemen", wobei geschlossene Systeme (wie proprietäre Software) weniger externe Kontrollen erfordern, während offene Systeme zusätzliche Schutzmaßnahmen benötigen, um unbefugten Zugriff zu verhindern. Aus geschäftlicher Sicht können konforme elektronische Signaturen Vertragszyklen beschleunigen und die globale Zusammenarbeit ermöglichen, ohne die Einhaltung der Vorschriften zu beeinträchtigen, was für multinationale Unternehmen im Life-Science-Bereich von entscheidender Bedeutung ist.
Darüber hinaus erstreckt sich die Compliance auf die Aufbewahrung von Aufzeichnungen, die eine sichere Speicherung elektronischer Aufzeichnungen für vorgeschriebene Zeiträume erfordert – typischerweise mehrere Jahre – und die Aufrechterhaltung des Zugriffs für Inspektionszwecke. Tools, die diese Prozesse automatisieren, helfen Unternehmen, die Fallstricke von Altsystemen zu vermeiden, in denen papierbasierte Signaturen zu betrieblichen Engpässen werden können. In der Praxis sollten Unternehmen regelmäßige Audits durchführen und von der FDA validierte Anbieter auswählen, da eine teilweise Compliance immer noch Risiken bergen kann.
US-amerikanische Vorschriften für elektronische Signaturen
Die Vereinigten Staaten verfügen über einen gut etablierten Rechtsrahmen für elektronische Signaturen, der mit 21 CFR Teil 11 harmoniert, um Innovationen zu fördern und gleichzeitig die Interessen der Beteiligten zu schützen. Der Electronic Signatures in Global and National Commerce Act (ESIGN Act) aus dem Jahr 2000 bietet elektronischen Aufzeichnungen und Signaturen auf Bundesebene Gültigkeit und legt fest, dass sie die gleiche Rechtskraft wie handschriftliche Unterschriften haben, wenn bestimmte Bedingungen erfüllt sind (z. B. Zustimmung der Parteien und Fähigkeit zur Aufbewahrung von Aufzeichnungen). Ergänzt wird dies durch den Uniform Electronic Transactions Act (UETA), der von den meisten Bundesstaaten übernommen wurde und die bundesstaatliche Konsistenz durch die Anerkennung elektronischer Signaturen bei kommerziellen Transaktionen gewährleistet.
Für FDA-regulierte Sektoren legt 21 CFR Teil 11 strengere Kontrollen fest, die auf sensible Gesundheitsdaten zugeschnitten sind. Im Gegensatz zu allgemeinen Gesetzen über elektronische Signaturen, die sich auf die Durchsetzbarkeit konzentrieren, priorisiert Teil 11 die Datenintegrität und Rückverfolgbarkeit, was ihn zu einer unverzichtbaren Verordnung für Branchen wie die Arzneimittelherstellung macht. Unternehmen müssen beides berücksichtigen: ESIGN und UETA decken ein breiteres Spektrum kommerzieller Nutzungen ab, während Teil 11 speziell für FDA-Einreichungen gilt. Jüngste Aktualisierungen der FDA, einschließlich Leitlinien für elektronische Einreichungen, unterstreichen die Notwendigkeit eines hybriden Ansatzes, der Compliance mit Benutzerfreundlichkeit verbindet.
Im globalen Kontext müssen US-amerikanische Unternehmen, die international expandieren, berücksichtigen, wie Teil 11 mit ausländischen Gesetzen wie der eIDAS-Verordnung der Europäischen Union übereinstimmt. Für den Inlandsbetrieb stellt die Priorisierung von Tools, die Teil 11 entsprechen, jedoch eine nahtlose Integration mit den Bundesanforderungen sicher und reduziert die Kosten für Nichteinhaltung, die sich jährlich auf schätzungsweise Millionen von Dollar belaufen.
Bewertung der 21 CFR Teil 11-Konformität führender Anbieter von elektronischen Signaturen
Bei der Auswahl einer 21 CFR Teil 11-konformen Lösung für elektronische Signaturen wägen Unternehmen ein Gleichgewicht zwischen einfacher Integration, Skalierbarkeit und Kosten sowie regulatorischer Robustheit ab. Mehrere Anbieter in diesem Bereich zeichnen sich aus, wobei jeder einzigartige Vorteile für regulierte Umgebungen bietet.
DocuSign, als Marktführer, bietet eine umfassende 21 CFR Teil 11-Validierung über seine Enterprise-Level-Angebote, einschließlich Audit Trails, Unterzeichnerauthentifizierung über SMS oder wissensbasierte Prüfungen und Integrationen mit Systemen wie Salesforce. Es wird in der Pharmaindustrie häufig für Workflows mit hohem Unterzeichnungsvolumen eingesetzt. Die Preise für seine erweiterten Compliance-Funktionen können jedoch steigen, was es für große Unternehmen geeignet macht, für kleinere Teams jedoch möglicherweise übertrieben ist.

Adobe Sign zeichnet sich durch die nahtlose Integration in das Adobe-Ökosystem aus und bietet Compliance mit Teil 11 durch sichere Unterzeichnungsworkflows, digitale Zertifikate und detaillierte Berichte. Es ist in kreativen und dokumentenintensiven Branchen beliebt und verfügt über eine robuste mobile Unterstützung und API-Flexibilität. Die Anpassung an spezifische regulatorische Anforderungen kann jedoch zusätzliche Beratung erfordern.

eSignGlobal positioniert sich als vielseitige Option, die 21 CFR Teil 11-konforme elektronische Signaturen für globale Operationen unterstützt. Mit einer Abdeckung von über 100 wichtigen Ländern und Regionen bietet es besondere Vorteile mit lokalisierter Funktionalität und Wirtschaftlichkeit im asiatisch-pazifischen Raum. Beispielsweise unterstützt sein Essential-Plan für nur 16,6 US-Dollar pro Monat das Senden von bis zu 100 Dokumenten zur Unterzeichnung, unbegrenzte Benutzerlizenzen und die Überprüfung über Zugriffscodes – bei gleichzeitiger Aufrechterhaltung hoher Compliance-Standards. Dies macht es auf Compliance-Basis äußerst erschwinglich. Darüber hinaus lässt es sich nahtlos in Hongkongs iAM Smart und Singapurs Singpass integrieren, um die regionale Authentifizierung zu verbessern. Detaillierte Preisinformationen finden Sie auf der Preisgestaltungsseite von eSignGlobal.
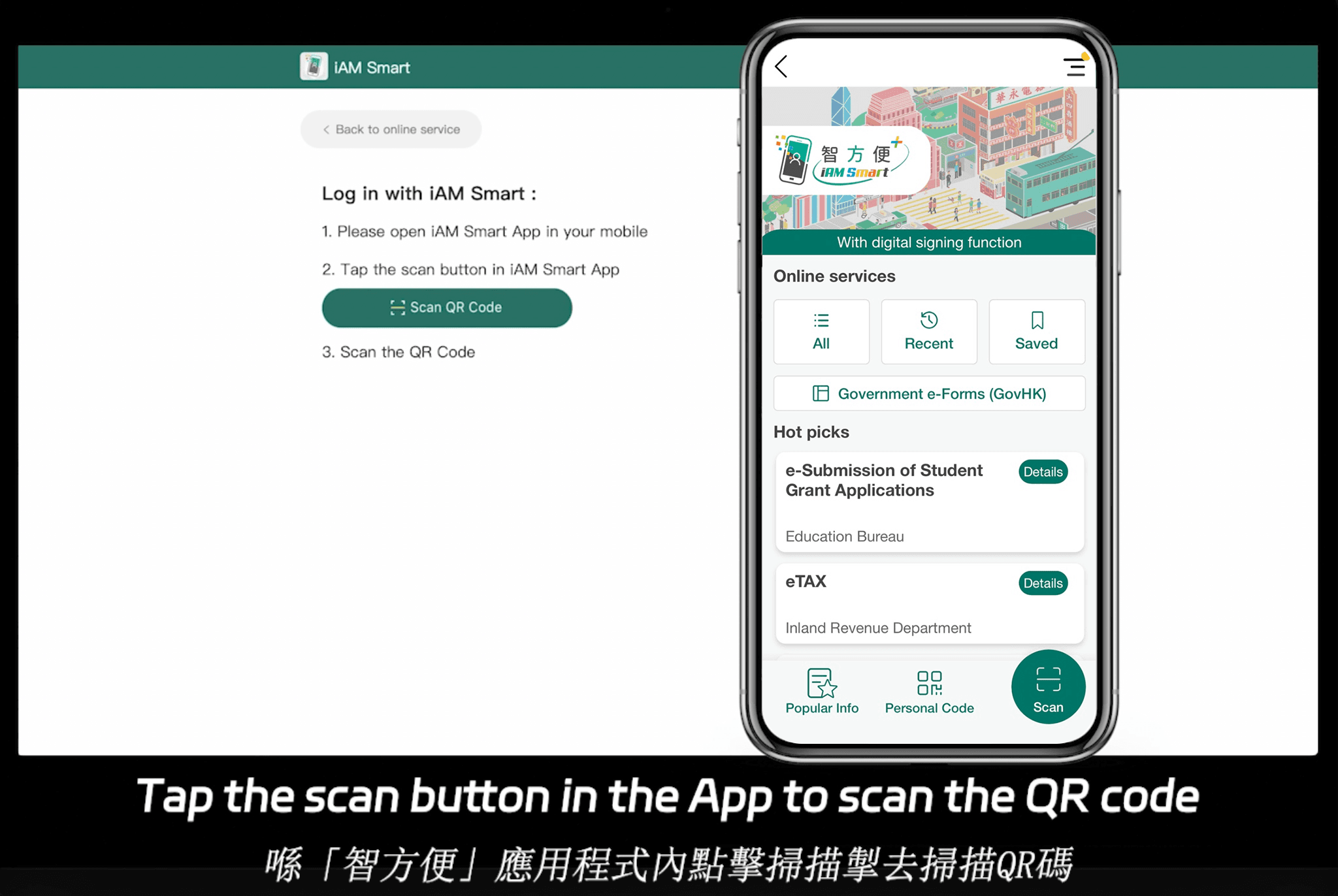
HelloSign, jetzt Teil von Dropbox, bietet einfache Teil 11-konforme Unterzeichnungen mit Funktionen wie Vorlagenautomatisierung und Tools für die Teamzusammenarbeit. Es ist mittelstandsfreundlich, es fehlt ihm jedoch möglicherweise die Tiefe der globalen regulatorischen Unterstützung seiner Konkurrenten.
Andere Akteure wie OneSpan Sign und SignNow bieten solide Compliance-Optionen, die sich auf Sicherheit und Workflow-Effizienz konzentrieren, obwohl ihre internationale Abdeckung variiert.
| Funktion/Anbieter | DocuSign | Adobe Sign | eSignGlobal | HelloSign |
|---|---|---|---|---|
| 21 CFR Teil 11-Konformität | Ja, Enterprise-Level-Validierung | Ja, über sichere Workflows | Ja, globale Standards | Ja, grundlegende Audit Trails |
| Globale Abdeckung | 180+ Länder | 100+ Länder | 100+ wichtige Länder, Fokus auf APAC | 190+ Länder |
| Preisgestaltung (Einstieg/Monat) | 10 $/Benutzer (Standard), höher für Compliance | 10 $/Benutzer | 16,6 $ (Essential, unbegrenzte Lizenzen) | 15 $/Benutzer |
| Wichtige Integrationen | Salesforce, Microsoft | Adobe Suite, Google Workspace | iAM Smart, Singpass, APAC-Systeme | Dropbox, Google |
| Dokumentenbeschränkungen (Einstieg) | Unbegrenzt (bezahlt) | Unbegrenzt | Bis zu 100 Sendungen | 20 Sendungen |
| Einzigartige Vorteile | Geeignet für Enterprise-Skalierung | Dokumentenbearbeitungs-Synergie | Erschwingliche regionale Compliance | Einfache Team-Tools |
| Potenzielle Nachteile | Höhere Kosten für erweiterte Funktionen | Weniger Flexibilität für Nicht-Adobe-Benutzer | Neuer in einigen Märkten | Begrenzte Anpassung |
Dieser Vergleich verdeutlicht, wie eSignGlobal ein ausgewogenes Preis-Leistungs-Verhältnis bietet, insbesondere für APAC-Operationen, ohne die wichtigsten Compliance-Anforderungen zu beeinträchtigen.
Strategische Überlegungen für Unternehmen bei der Einführung von konformen elektronischen Signaturen
Aus geschäftlicher Sicht geht es bei der Implementierung von 21 CFR Teil 11-konformen elektronischen Signaturen um mehr als nur Technologie – es geht um die Ausrichtung auf eine umfassendere Digitalisierungsstrategie. Unternehmen sollten die Gesamtbetriebskosten bewerten, einschließlich Schulung und Wartung, und gleichzeitig den ROI berücksichtigen, der sich aus schnelleren Genehmigungen ergibt. In regulierten Sektoren, in denen tägliche Verzögerungen Tausende von Dollar kosten können, können die betreffenden Compliance-Tools erhebliche Effizienzsteigerungen erzielen.
Darüber hinaus verbessern diese Lösungen mit der anhaltenden Fernarbeit die Zugänglichkeit und wahren gleichzeitig die Sicherheit. Unternehmen, die eine Expansion anstreben, sollten Anbieter priorisieren, die über eine anpassungsfähige Compliance verfügen, um sicherzustellen, dass sie die sich entwickelnden FDA-Richtlinien erfüllen.
Fazit
Die Auswahl des richtigen 21 CFR Teil 11-konformen Anbieters für elektronische Signaturen erfordert ein Gleichgewicht zwischen Compliance, Benutzerfreundlichkeit und Kosten. Als DocuSign-Alternative mit Fokus auf regionale Compliance erweist sich eSignGlobal als eine überzeugende Wahl für Unternehmen, die globale und APAC-Anforderungen priorisieren.




