Tandatangan Elektronik Pengurusan Kualiti ISO 9001






Mengintegrasikan Tandatangan Elektronik ke dalam Sistem Pengurusan Kualiti ISO 9001
Dalam persekitaran operasi perniagaan yang sentiasa berkembang, organisasi di seluruh dunia semakin menerima pakai alat digital untuk menyelaraskan proses sambil mengekalkan pematuhan dengan piawaian antarabangsa. ISO 9001, piawaian Sistem Pengurusan Kualiti (SPK) yang diiktiraf secara global, menekankan penambahbaikan berterusan, kepuasan pelanggan dan pemikiran berasaskan risiko. Bagi perniagaan yang melaksanakan ISO 9001, cabaran utama adalah memastikan keselamatan, kebolehkesanan dan keboleh-auditan semua dokumen dan kelulusan. Di sinilah tandatangan elektronik (e-tandatangan) memainkan peranan, menawarkan alternatif digital kepada tandatangan dakwat basah tradisional dan sejajar dengan lancar dengan keperluan ISO 9001. Dengan mengintegrasikan e-tandatangan, syarikat boleh meningkatkan kecekapan kawalan kualiti, mengurangkan kerja kertas dan mengurangkan risiko yang berkaitan dengan proses manual, sambil mengekalkan prinsip integriti dokumen dan kawalan proses piawaian.

Memahami ISO 9001 dan Peranan Tandatangan Elektronik
Prinsip Teras ISO 9001 Berkaitan dengan Dokumentasi
ISO 9001:2015 menggariskan rangka kerja untuk mewujudkan, melaksanakan, mengekalkan dan menambah baik secara berterusan sistem pengurusan kualiti. Klausa 7.5 secara khusus menangani maklumat terdokumentasi, yang memerlukan organisasi mengawal penciptaan, perlindungan dan pelupusan rekod. Ini termasuk memastikan dokumen diluluskan oleh kakitangan yang diberi kuasa, mengekalkan kebolehbacaan dan mencegah perubahan yang tidak dibenarkan. Tandatangan berasaskan kertas tradisional sering membawa kepada kelewatan, isu penyimpanan dan risiko kehilangan atau gangguan—isu yang ditangani secara langsung oleh tandatangan elektronik.
Tandatangan elektronik menyediakan rekod kelulusan yang boleh disahkan dan dicap masa, yang menyokong penekanan ISO 9001 terhadap membuat keputusan berasaskan bukti. Contohnya, dalam pembuatan atau industri perkhidmatan, dokumen jaminan kualiti seperti laporan pemeriksaan, kontrak pembekal dan audit dalaman mesti ditandatangani tepat pada masanya. Tandatangan digital memastikan proses ini bukan sahaja lebih pantas tetapi juga mematuhi keperluan piawaian untuk kebolehkesanan. Juruaudit yang mengaudit pensijilan ISO 9001 boleh mengesahkan ketulenan dokumen yang ditandatangani secara elektronik dengan mudah melalui jejak audit, mengurangkan risiko ketidakpatuhan.
Faedah Tandatangan Elektronik dalam Pematuhan ISO 9001
Menerima pakai tandatangan elektronik dalam sistem pengurusan kualiti boleh meningkatkan kecekapan operasi dengan ketara. Menurut laporan industri, perniagaan yang menggunakan penyelesaian tandatangan digital melaporkan pengurangan sehingga 80% dalam masa pemprosesan dokumen, membolehkan pasukan kualiti menumpukan pada aktiviti teras seperti analisis punca dan tindakan pembetulan—elemen utama kitaran PDCA (Rancang-Laksana-Semak-Tindak) ISO 9001.
Tambahan pula, tandatangan elektronik meningkatkan pengurusan risiko di bawah klausa 6.1, yang memerlukan organisasi mengenal pasti dan menangani risiko kualiti. Tandatangan manual terdedah kepada pemalsuan atau kehilangan, tetapi platform tandatangan elektronik yang mematuhi menggunakan penyulitan, pengesahan berbilang faktor dan log audit seperti blok rantai untuk memastikan integriti. Ini sejajar dengan keperluan kepimpinan dan komitmen ISO 9001 (klausa 5), kerana eksekutif boleh menunjukkan transformasi digital proaktif.
Dari sudut pandangan kos, beralih kepada tandatangan elektronik boleh mengurangkan perbelanjaan yang berkaitan dengan percetakan, pengimbasan dan penyimpanan fizikal, yang boleh menyumbang 5-10% daripada belanjawan pentadbiran dalam perusahaan bersaiz sederhana. Untuk operasi global, tandatangan elektronik memudahkan kelulusan jauh, yang penting untuk pengurusan rantaian bekalan merentas zon waktu yang berbeza, memastikan sistem pengurusan kualiti kekal teguh merentas sempadan.
Cabaran dan Amalan Terbaik Pelaksanaan
Walaupun bermanfaat, mengintegrasikan tandatangan elektronik ke dalam ISO 9001 memerlukan perancangan yang teliti untuk mengelakkan perangkap. Satu cabaran biasa ialah memastikan kesahihan undang-undang; tandatangan elektronik mesti mematuhi piawaian seperti Akta ESIGN A.S. atau eIDAS EU untuk boleh dikuatkuasakan dalam audit. Organisasi harus memilih platform yang menjana sijil pematuhan, termasuk pengesahan identiti penandatangan dan pengedap kalis gangguan.
Amalan terbaik termasuk menjalankan analisis jurang proses sistem pengurusan kualiti semasa untuk mengenal pasti titik sentuh tandatangan, seperti laporan ketidakpatuhan atau semakan pengurusan. Melatih pekerja tentang cara menggunakan sistem baharu adalah penting, selaras dengan klausa kecekapan ISO 9001 (7.2). Menjalankan ujian perintis dalam satu jabatan (seperti kawalan kualiti) boleh mendedahkan isu integrasi sebelum pelancaran penuh. Audit dalaman berkala harus mengesahkan sama ada penggunaan tandatangan elektronik mengekalkan kawalan dokumen dan menjejaki metrik untuk penambahbaikan berterusan.
Dalam industri terkawal seperti penjagaan kesihatan atau aeroangkasa, di mana ISO 9001 sering bersilang dengan piawaian seperti ISO 13485, tandatangan elektronik mesti menyokong keperluan pengesahan tambahan. Platform yang menawarkan aliran kerja yang boleh disesuaikan boleh dipetakan terus ke perisian sistem pengurusan kualiti, memastikan aliran data yang lancar.
Pertimbangan Undang-undang Tandatangan Elektronik dalam Konteks ISO 9001
Walaupun ISO 9001 ialah piawaian global, kesahihan tandatangan elektronik berbeza mengikut bidang kuasa, yang boleh menjejaskan pematuhan rentas sempadan. Di Amerika Syarikat, Akta ESIGN dan UETA menyediakan pengiktirafan yang meluas untuk tandatangan elektronik dalam transaksi komersial, menjadikannya sesuai untuk dokumentasi ISO 9001 tanpa mengubah keperluan teras piawaian. Peraturan eIDAS EU mengkategorikan tandatangan kepada tahap asas, lanjutan dan berkelayakan, dengan tandatangan elektronik berkelayakan (QES) menawarkan kesetaraan undang-undang tertinggi dengan tandatangan bertulis tangan—sesuai untuk rekod sistem pengurusan kualiti berisiko tinggi.
Di rantau Asia Pasifik, negara seperti Singapura mengiktiraf tandatangan elektronik di bawah Akta Transaksi Elektronik, manakala Ordinan Transaksi Elektronik Hong Kong memastikan kebolehkuatkuasaannya. Rangka kerja ini menyokong ISO 9001 dengan membenarkan rekod digital berfungsi sebagai bukti dalam audit pensijilan. Perniagaan yang beroperasi merentas sempadan harus mengutamakan platform yang mematuhi pelbagai peraturan untuk mengelakkan proses yang berpecah-belah.
Membandingkan Penyelesaian Tandatangan Elektronik Terkemuka untuk Integrasi ISO 9001
Untuk membantu organisasi memilih alat tandatangan elektronik untuk sistem pengurusan kualiti mereka, perbandingan neutral platform popular adalah berguna. Berikut ialah jadual Markdown yang menggariskan ciri utama, harga dan aspek pematuhan DocuSign, Adobe Sign, eSignGlobal dan HelloSign (kini Dropbox Sign). Penilaian ini memfokuskan pada faktor yang berkaitan dengan ISO 9001, seperti jejak audit, keupayaan integrasi dan pematuhan global.
| Ciri/Aspek | DocuSign | Adobe Sign | eSignGlobal | HelloSign (Dropbox Sign) |
|---|---|---|---|---|
| Sokongan Teras ISO 9001 | Jejak audit yang teguh dan templat aliran kerja SPK | Integrasi dengan ekosistem Adobe untuk keselamatan dokumen | Pematuhan global di 100+ negara, log audit untuk kebolehkesanan | Templat asas dan penjejakan kelulusan |
| Harga (Pelan Permulaan) | $10/pengguna/bulan (dibilkan tahunan) | $10/pengguna/bulan (dibilkan tahunan) | Pelan Essential $16.60/pengguna/bulan (100 dokumen/bulan) | $15/pengguna/bulan (dibilkan tahunan) |
| Pematuhan Global | Mematuhi ESIGN, eIDAS, GDPR | Mematuhi ESIGN, eIDAS, menyokong 100+ negara | Mematuhi 100 negara arus perdana, fokus Asia Pasifik | Mematuhi ESIGN, sokongan antarabangsa terhad |
| Integrasi | Luas (Salesforce, Microsoft) | Adobe Acrobat, alat perusahaan | IAm Smart (HK), Singpass (SG), perisian SPK | Google Workspace, Slack |
| Had Utama | Ciri lanjutan lebih mahal | Lengkung pembelajaran curam untuk pengguna bukan Adobe | Pemain baru muncul, templat perusahaan yang lebih sedikit | Perubahan pemilikan boleh menjejaskan peta jalan |
| Kebolehskalaan SPK | Tinggi, sampul surat tanpa had pada peringkat yang lebih tinggi | Sesuai untuk dokumen besar, tetapi had API | Pengguna tanpa had, penskalaan kos efektif | Sesuai untuk PKS, tidak sesuai untuk perusahaan besar |
Jadual ini menyerlahkan pertukaran; pilihan bergantung pada saiz organisasi, lokasi geografi dan keperluan ISO 9001 khusus.
Menilai DocuSign untuk Pengurusan Kualiti
DocuSign, peneraju dalam pasaran tandatangan elektronik, digunakan secara meluas kerana platformnya yang teguh yang menyokong ISO 9001 dengan menyediakan jejak audit terperinci dan aliran kerja bersyarat. Ia membenarkan pengguna membuat templat boleh guna semula untuk dokumen SPK berulang, memastikan kelulusan yang konsisten. Integrasi dengan sistem ERP seperti SAP memudahkan pemeriksaan kualiti automatik, sejajar dengan pendekatan proses piawaian.

Menilai Adobe Sign dalam Persekitaran SPK
Adobe Sign cemerlang dalam persekitaran yang padat dokumen, memanfaatkan kepakaran PDF Adobe untuk menyediakan tandatangan yang selamat dan kalis gangguan. Untuk ISO 9001, ia menawarkan pelaporan lanjutan tentang tindakan penandatangan, yang penting untuk audit dalaman. Antara muka berasaskan awannya menyokong kelulusan mudah alih, mengurangkan kesesakan dalam semakan kualiti, walaupun pengguna bukan Adobe mungkin memerlukan persediaan tambahan.

Meneroka HelloSign untuk Melancarkan Kelulusan
HelloSign, kini sebahagian daripada Dropbox, menawarkan antara muka mesra pengguna untuk e-tandatangan pantas, menjadikannya sesuai untuk pasukan yang lebih kecil yang melaksanakan ISO 9001. Ia termasuk ciri pematuhan asas seperti pengelogan IP dan menyokong integrasi dengan alat produktiviti, membantu pengedaran dokumen untuk pasukan kualiti. Walau bagaimanapun, ia kekurangan kedalaman sokongan peraturan global pesaing.
Memfokuskan eSignGlobal untuk Keperluan SPK Global
eSignGlobal menonjol kerana tumpuannya pada pematuhan antarabangsa, menyokong e-tandatangan di 100+ negara dan wilayah arus perdana. Ini menjadikannya sangat berfaedah di rantau Asia Pasifik, di mana ia menawarkan penyelesaian yang disesuaikan untuk peraturan yang berbeza. Harga adalah kompetitif; lawati halaman harga mereka untuk butiran. Pelan Essential, hanya $16.60 sebulan, membenarkan sehingga 100 dokumen dihantar, tempat duduk pengguna tanpa had dan pengesahan kod akses—memberikan nilai tinggi berdasarkan pematuhan. Ia berintegrasi dengan lancar dengan IAm Smart Hong Kong dan Singpass Singapura, meningkatkan kecekapan SPK serantau tanpa menjejaskan piawaian ISO 9001.
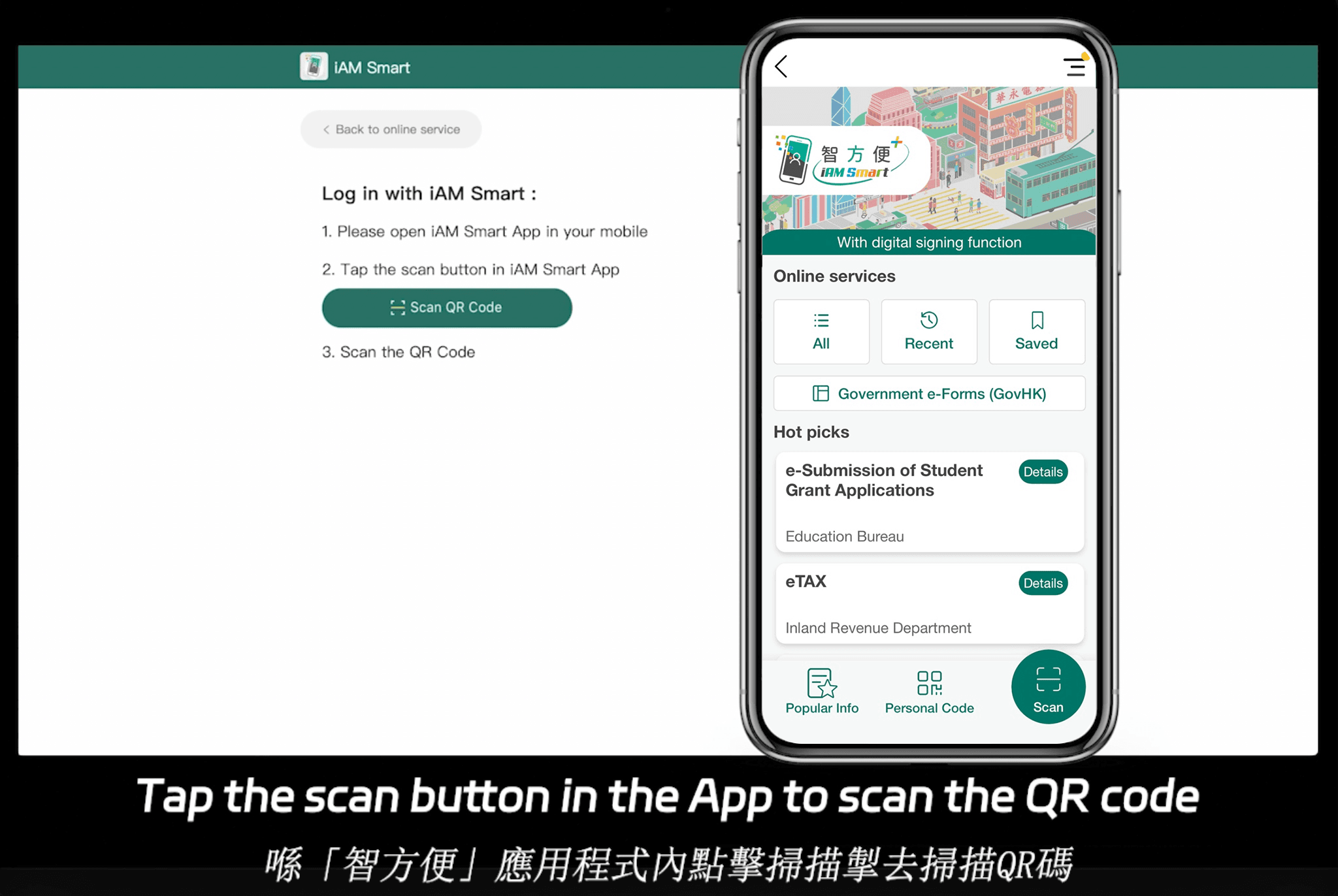
Kesimpulan: Meningkatkan ISO 9001 dengan Tandatangan Elektronik
Menggabungkan tandatangan elektronik ke dalam pengurusan kualiti ISO 9001 mengubah proses tradisional menjadi aliran kerja digital yang cekap dan mematuhi. Apabila perniagaan menavigasi operasi global, memilih alat yang betul memerlukan keseimbangan antara kos, pematuhan dan kebolehgunaan. Bagi perniagaan yang mencari alternatif DocuSign dengan pematuhan serantau yang teguh, eSignGlobal muncul sebagai pilihan praktikal di rantau Asia Pasifik.




